| на русском | in english | ||||||

|

|

|

|

|

|  |
About drug dosage
The traditional approach of a choice of drug dosage
The traditional approach assume use of pharmacological models of the anesthetics in the form of recommended doses and frequencies of their administration.
The dose corresponds to a desirable therapeutic range at regular administration of a drug.
The dose size is defined by the "average" value depending on change of drug concentration in a therapeutic range (Jambhekar S.S., Breen P.J., 2009).
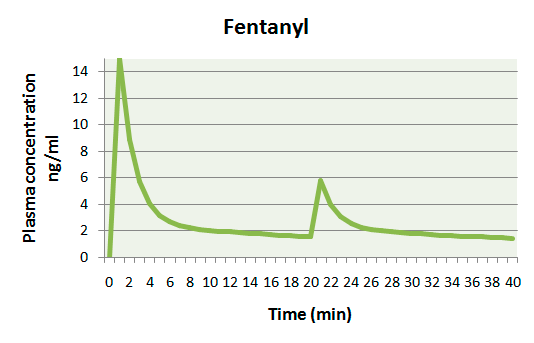 The picture (CRITICAL.RU)
on the right is shown the example of change of fentanyl plasma concentration after administration bolus 5 mkg/kg and repeated bolus 100 mkg in 20 minutes.
According to the literature (Miller R.D., 2009) the bottom threshold of analgetic concentration of fentanyl is 2 ng/ml.
In the picture it is shown that at traditional administration of fentanyl from 8th minute and to the following bolus of drug fentanyl concentration corresponds inadequate analgesia during surgery.
The picture (CRITICAL.RU)
on the right is shown the example of change of fentanyl plasma concentration after administration bolus 5 mkg/kg and repeated bolus 100 mkg in 20 minutes.
According to the literature (Miller R.D., 2009) the bottom threshold of analgetic concentration of fentanyl is 2 ng/ml.
In the picture it is shown that at traditional administration of fentanyl from 8th minute and to the following bolus of drug fentanyl concentration corresponds inadequate analgesia during surgery.
The similar approach allows to choose induction doses, but doesn't give quantitative and time reference points for a substantiation of anesthesia maintenance. The simplified and inexact pharmacokinetics of a drug based on "average" anesthetic concentration, leads to that possible calculation of a total dose of an administered drugs for a time unit (minute, hour) becomes not informative. Introduction of maintenance doses with orientation to clinical signs (changes blood pressure, heart rate and others) without assessment current levels of drugs concentrations becomes insufficiently proved. Besides, approximateness of the traditional approach doesn't give possibility for the numerical analysis of current plasma concentration of anesthetic and its comparison to drug action.
Calculated plasma concentration is the best choice
The existence of pharmacological models of the drugs used during general anesthesia, allows to calculate concentrations of these drugs for any interested physician point in time.
The similar approach allows to analyze the total administered dose, tactics of administration of parts making it
and also necessary level of drug concentration.
As parameters of pharmacological models are calculated for certain groups of volunteers taking into account their anthopometrical, sexual, age and other parameters,
the wide choice of models allows the physician to analyze the change concentration of any drugs depending on the selected group.
PK/PD compartmental models
For the description of pharmacokinetics of drugs are used compartmental models.
These are the mathematical models representing simplified schemes of elimination and redistribution of drugs in compartments.
Each model defines quantity of compartments and their volumes, speed of a metabolism, elimination of drugs
and speed of redistribution between compartments.
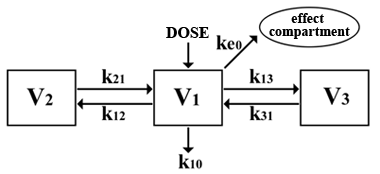
The compartment into which the drug is entered is called as central (V1);
second and third – peripheral compartments.
Compartments of models are theoretical and have no real anatomo-physiological communications.
Speed of a metabolism and drug distribution is described by constants of speed(k) (Absalom A.R., 2007).
Effect-site concentration
After drug administration the short latent period between increase plasma concentration and occurrence of clinical effect is observed,
and after the termination of infusion concentration falling precedes decrease in effect of the administered drug.
This results from the fact that the clinical effect is defined by concentration of the anesthetics not in blood plasma, but in effect-site.
For the best interpretation of pharmacokinetics and pharmadynamics of the anesthetics,
to pharmacological model is added the "effect compartment" which reflects drugs concentration in effect site(Ce).
Speed of movement of a drug in this compartment and its removal is described only by one constant which is usually designated as ke0.
As drug concentration can't be measured in a target organ directly calculation Ce is carried out on the basis of a constant ke0
which reflects time before achievement of peak anesthetic concentration in effect compartment.
This constant is established by measurement of clinical effect, for example, by means of electroencephalography.
Accuracy of PK/PD models
For check of accuracy of pharmacological model are conducted researches in which compare measured concentration of
drugs in tests of blood and calculated plasma concentration in corresponding time points.
Most often used criteria for an estimation of accuracy of pharmacological models include MDPE
(median performance error), MDAPE (median absolute performance error) (Varvel J.R. et al., 1992).
For pharmacological models values are satisfactory MDPE up to 10-20% and MDAPE up to 20-40% (Schuttler J. et al., 1988; Glass P.J.A. et al., 1990).
It is comparable to accuracy of measurement of end-tidial concentration of the inhalation anesthetics.
So, end-tidial isoflurane concentration differs from measured in arterial blood on 20 %(Frei F.J. et al., 1991; Dwyer R.C. et al., 1991).
Most often used PK/PD models correspond to the specified norms on accuracy.
However the deviation of settlement concentration from real is supposed owing to massive intraoperative blood loss,
large volume of intravenous infusion.
For the decision of this problem expansion of indicators for monitoring to achieve of adequate feedback is required.
Possibility to analyze plasma concentration of all used drugs during surgery is one more direction of monitoring which allows to compare
current plasma concentration of drugs with actions of surgeons at present time, parameters of hemodynamic, EEG, ECG and with any other actively changing indicators.
There are recommendations about a choice and maintaince of drugs plasma concentration during various stages of surgery
which are a basis for selection of tactics of administration drugs.
What is recomendations? 
1.Absalom A.R., Struys R.F. An Overview of TCI and TIVA. Belgium: Gent, Academia Press, 2007. – 108 p.
2.Dwyer R.C., Fee J.P., Howard P.J., Clarke R.S. Arterial washin of halothane and isoflurane in young and eldery adult patients. Br. J. Anaesth. – 1991. – Vol. 66. – P. 572–579.
3.Frei F.J., Zbinden A.M., Thomson D.A., Rieder H.U. Is the end–tidal partial pressure of isoflurane a good predictor of its arterial partial pressure? Br. J. Anaesth. – 1991. – Vol. 66. – P. 331–339.
4.Glass P.S., Jacobs J.R., Smith L.R. et al. Pharmacokinetic model–driven infusion of fentanyl: Assessment of accuracy. Anesthesiology. 1990. – Vol. 73. – P. 1082–1090.
5.Hull C.J., Beem H.B.H., McLeon K. et al. A pharmacodynamic model for pancuronium. Br. J. Anaesth. – 1978. – Vol. 50. – P. 1113–1123.
6.Jambhekar S.S., Breen P.J. Basic Pharmacokinetics, Pharmaceutical Press, 2009, p. 425
7.Miller R.D. Miller's anesthesia. 7th edition. 2009. Раздел III: Anesthetic Pharmacology.
8.Schuttler J., Kloos S., Schwilden H., Stoeckel H. Total intravenous anaesthesia with propofol and alfentanil be computer–assisted infusion. Anaesthesia. – 1988. – Vol. 43. – P. 2–7.
9.Varvel J.R., Donoho D.L., Shafer S.L. Measuring the predictive performance of computer–controlled infusion pumps. J. Pharmacokinet. Biopharm. – 1992. – Vol. 20. – P. 63–94.
| ANAESTHSOFTWARE.COM
| ||